|
Millikan's method involved watching the behavior of oil droplets in an electrically charged field. 
" It was still at least possible that electrons came in a spectrum of charges. Moreover, until one had a relatively direct way of measuring the charge on the electron, one couldn't really be sure that the electron was indeed an "atom of electricity. Around the turn of the century, Thomson and several of his students at the Cavendish Laboratory tried various means of indirect measurement, with unconvincing results. But neither, it seemed, could you isolate an electron and measure its charge on an electrometer. Measuring the mass of an electron seemed out of the question you couldn't put one on a scale and read a dial. But without knowing either the charge e or the mass m of the electron, all one had was a set of relative values it would be like knowing a set of values in units of a foreign currency - that a house costs x times what a car costs, which in turn costs y times what a newspaper costs - without knowing the value in your own currency of any one. The e/m ratio of the electron was related to a number of other important quantities of interest to scientists. Thomson had discovered the electron and measured the ratio of its charge to mass (the e/m ratio) - an event which helped to usher in the electronic age. He had won the Nobel Prize in 1924, largely due to his important and innovative measurement, carried out around 1910, of the charge on the electron - one of the most central physical constants that scientists of that era had been seeking to determine. 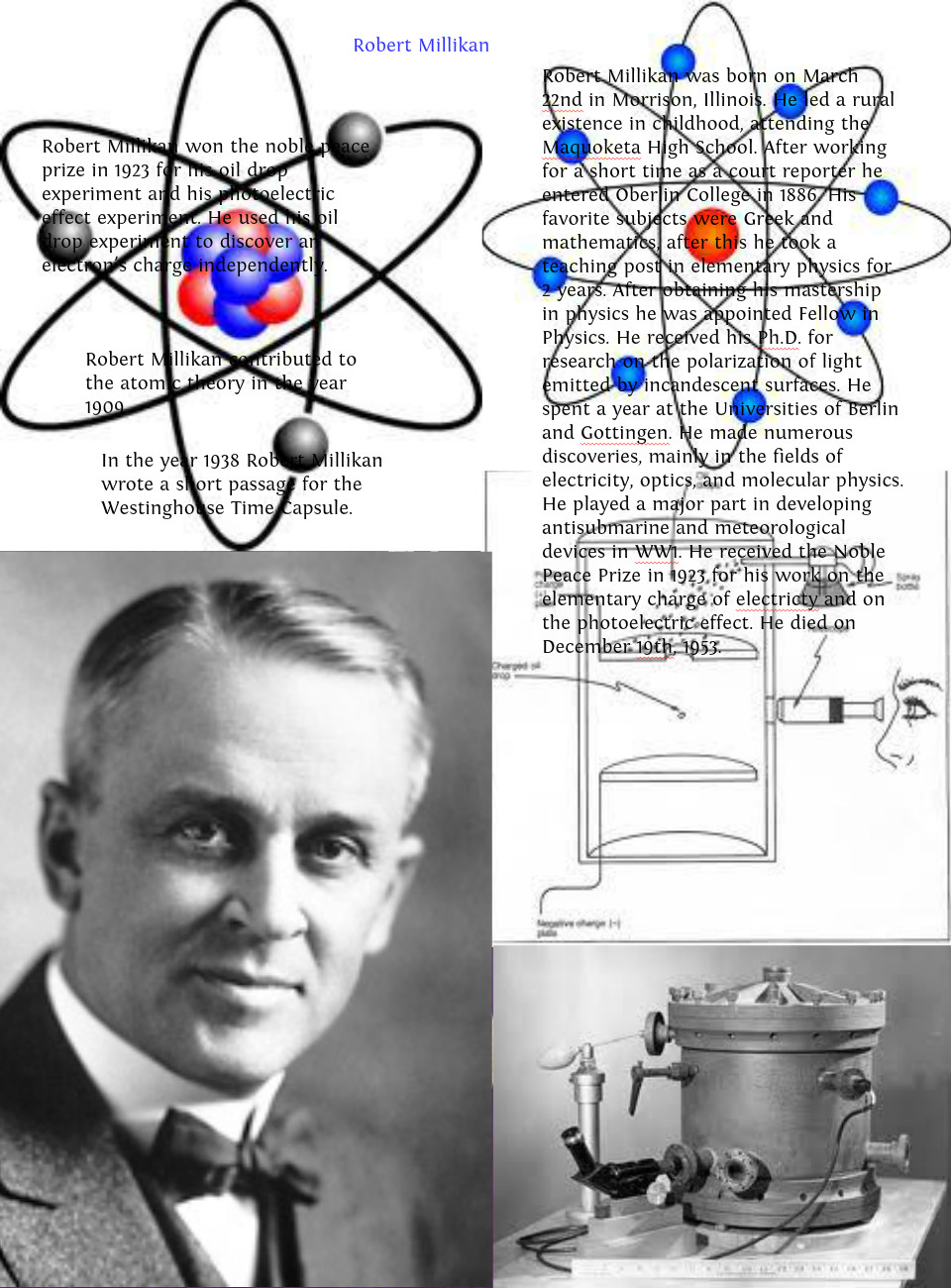
An acceptable practice, but one that is rarely followed, is to decide in advance what specific observed circumstances in a particular experimental situation would justify data rejection. In some cases statistical rules can be used as guidance, but in many situations it is left to the judgement of the experimental scientist to decide if a problem with the equipment or some other consideration justifies discarding a datum or a set of data. Scientists agree that there are circumstances when some of the data collected in an experiment can be rejected or disregarded. As those historians who have analyzed the record of Millikan's treatment of his experimental data, and other similar cases, have pointed out, it is not always easy to distinguish between the "right" thing to do, inappropriate but inadvertent manipulation, and intentional fudging. One can, however, set up simpler situations to illustrate the same points. But by itself, it is probably too complicated to use in helping students to navigate this issue. The Millikan case highlights a number of the important issues involved. What makes this an ethical issue, rather than just an issue of laboratory practice, is that the action that most promotes one's self-interest can be different from the "right" thing to do. It happens all the time: their instruments give one result - but they know that everybody else is getting some other result, or that something's wrong with the way they have done it, or that the result that people got last year is different, or that the result in the book is different. In 1904, Thomson proposed the “plum pudding” model of atoms, which described a positively charged mass with an equal amount of negative charge in the form of electrons embedded in it, since all atoms are electrically neutral.Of all the various types of "ethical dilemmas in science," the temptation to "fudge", or even invent data outright, is probably the one which high school science students will find most familiar. However, the positively charged part of an atom was not yet well understood. Scientists had now established that the atom was not indivisible as Dalton had believed, and due to the work of Thomson, Millikan, and others, the charge and mass of the negative, subatomic particles-the electrons-were known. Since the charge of an electron was now known due to Millikan’s research, and the charge-to-mass ratio was already known due to Thomson’s research (1.759 10 11 C/kg), it only required a simple calculation to determine the mass of the electron as well. 
Millikan concluded that this value must therefore be a fundamental charge-the charge of a single electron-with his measured charges due to an excess of one electron (1 times 1.6 10 −19 C), two electrons (2 times 1.6 10 −19 C), three electrons (3 times 1.6 10 −19 C), and so on, on a given oil droplet. Looking at the charge data that Millikan gathered, you may have recognized that the charge of an oil droplet is always a multiple of a specific charge, 1.6 10 −19 C. The tabulated data are examples of a few possible values. 
\): Millikan’s experiment measured the charge of individual oil drops.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
